Posts.
Field notes from the front of synthetic dye reformulation. Every post is a real bench observation on natural color behavior, stability, and the 2027 phase-out, published as it happened. The full text of each lives here, indexed and citable.

A bomb pop isn't one product
A bomb pop isn't one product. It's three products stacked. Each layer has its own pH, its own sugar profile, its own freeze curve. When you formulate with synthetic dyes, those layers can ignore each other. Red 40 doesn't care what the layer underneath it is doing. Blue 1 doesn't blink at a frozen acid bath. Natural colors don't have that luxury. Anthocyanins shift hue with every tenth of a pH point. Acidic = red. Neutral = purple. Alkaline = blue-gray. Spirulina and butterfly pea blue both have narrow pH windows and degrade under thermal cycling. The kind of cycling a grocery freezer does every time the door opens. Now stack them. The red layer bleeds a fraction of its acid pH into the white. White's pH shifts. The blue underneath, already borderline, gets dragged off-spec. By the time the consumer gets it home, your bomb pop bombed. The fix isn't picking a more stable red. It's modeling the whole stack. pH per layer, freeze curve per layer, thermal-cycle exposure across shelf life. Before you commit to any single ingredient. That's the multi-variable trap DyeConverter(TM) was built for. The platform models pH, thermal stability, freeze-cycle exposure, and ingredient interaction so you can see which naturals play nicely together BEFORE you spin a batch. Sometimes adding a color doesn't just break itself. It breaks the one next to it. #FutureOfFood #DyeConverter #FDA2027

Sequence Matters
Sequence Matters: The donut, the sports drink, and the cereal aren't solving the same problem. The donut's pink is a surface application. Frosting meets dye after the bake. Short heat exposure, low water activity, and dye sits on top, not in. The reformulator worries about adhesion, freeze-thaw cycles in distribution, and fade under case lights. The sports drink's red is fully dissolved in a low-pH liquid. Citric or lactic acid. Exposed to fluorescent and sun for 18 months on the shelf. The reformulator worries about anthocyanin pH stability, oxidation, light degradation, and mineral chelation. The cereal's stars are dyed at over 300°F during extrusion or surface-coated after. Then they sit dry for a year or more. The reformulator worries about thermal degradation, crumb-color migration, oxidation, and texture. Same FDA mandate. Same color on the label. Three completely different sequences of chemistry to solve. This is what makes the 2027 timeline so deceptive. Brands plan as if "reformulation" is one project. It isn't. It's a different project per category, per matrix, and per shelf-life ask. DyeConverter™ was built to hold all of those sequences at once. The donut sequence. The beverage sequence. The cereal sequence. Each with its own pH window, thermal limit, candidate ingredient set, and regulatory map. Reformulation is not a single event. It's a portfolio of them. #CleanColor #DyeConverter #FDA2027

Why Did My Salmon Turn Pink?
Why Did My Salmon Turn Pink? Salmon gets its color from astaxanthin, a lipid-soluble carotenoid that is bound within the muscle tissue. It is integrated into the protein structure and produces a stable, muted orange-pink. Try to replicate that system with a natural, water-soluble pigment? It might end up looking radioactive. Natural reds like beets (betalains) are not protein-bound. They sit in the aqueous phase, reflect light at the surface, and can become highly saturated when overdosed. The result is a color that looks glossy, uniform, and artificially bright instead of structurally integrated. Same target color. Completely different chemistry. This is where reformulations break down. Swapping artificial dyes with natural ones is not a 1:1 match. Natural dyes are fickle, fragile, and finicky. Replacing a pigment without accounting for solubility, binding behavior, and matrix interaction will not produce the same visual outcome. And time is money. But right now, you don't have time for expensive trial and error. That's why I built DyeConverter: to help you find the perfect natural dye replacement based on pH, process, and protein values. We're not a manufacturer; we're not a marketplace. We're simply the matchmaker. Because you've got better things to do than endless researching and calling for samples. You've got an entire portfolio to replace... Stop researching; stop guessing. Get to market faster.

De-risk your reformulations
De-risk your reformulations The one on the left? Natural dye. The one on the right? Also natural dye. The risk isn't deciding to switch from synthetic; it's finding a SKU that actually performs in your product. I built DyeConverter™ to be more than a platform: it's an AI-powered decision model that knows how fragile natural dyes can be. Tell it your current FD&C color, and it recommends the right natural match based on your formulation, process, and packaging needs. (Parts per million included.) Your time has never been more valuable. De-risk your decision with DyeConverter and get to market faster. #CleanColor #DyeConverter #PPM #ArtificialDyes

Why did my blue hard candy turn purple the second I added citric acid?
Why did my blue hard candy turn purple the second I added citric acid? Because butterfly pea flower is not Blue 2. It is an anthocyanin. And anthocyanins are pH chameleons. Between pH 3.8 and 7, you get that beautiful natural blue. Drop citric acid or lemon juice into the mix and the pH crashes. The pigment shifts to vivid purple right in front of you. Here is the thing most R&D teams miss. That purple is not a defect. It is actually a unique shade you cannot replicate with synthetic FD&C dyes. Some formulators are leaning into it on purpose to create sour candy and confections with a color profile that stands out on shelf. The trick is process control. Add butterfly pea flower at the very end of your heating cycle. Less thermal stress means better color retention, lower usage rates, and more stability through shelf life. This is one of the formulation patterns DyeConverter™ maps automatically. Input your application, pH, and processing conditions. Get back the alternatives that actually work for your specific product. #NaturalColorants #DyeConverter #FoodScience

Why is my lemon-lime soda turning green when I add spirulina?
Why is my lemon-lime soda turning green when I add spirulina? Because spirulina is not yellow. It is blue-green. And when you drop a blue-green pigment into a citric acid beverage expecting it to replace Yellow 5, you get swamp water. This is one of the most common mistakes in natural color reformulation. Teams reach for spirulina because it is familiar and approved. But phycocyanin, the pigment in spirulina, absorbs red light and reflects blue. Mix that with a yellow base and the math gives you green every time. The fix is not spirulina at all. For a lemon-lime soda, you need a true yellow source. Safflower. Turmeric with proper encapsulation. Beta-carotene emulsions. Each one behaves differently at low pH, under carbonation pressure, and across shelf life. This is exactly the kind of application-specific logic that DyeConverter™ maps. You input the product type, pH, processing conditions, and target shade. The platform returns alternatives ranked by stability, regulatory status, and cost -- so your R&D team is not guessing their way through a reformulation that has a hard deadline. The 2027 clock is running. The wrong pigment in the wrong matrix costs months you do not have. #NaturalColorants #FoodFormulation #DyeConverter

Why does spirulina fade in gummies?
Why does spirulina fade in gummies? Short answer: the proteins are breaking down before the gummy hits the bag. Spirulina blue comes from phycocyanin. It's a protein-based pigment. And like all proteins, it has a narrow window where it stays intact. Step outside that window with too much heat or too low a pH and the protein denatures. That's your color disappearing. Here's where most teams get stuck. They add spirulina to the candy mass while it's still hot. The phycocyanin hits 180F+ and starts falling apart immediately. By packaging, the color is spotty, faded, or shifted toward green. The fix is timing. Add spirulina during the cooling stage, around 170F (77C). High enough for workability. Low enough to keep the protein intact. And if your formula includes citric acid, delay that addition until after the spirulina is fully incorporated. Acid and heat together will destroy the color faster than either one alone. This is one of 65 formulation challenges mapped inside DyeConverter. The platform matches natural alternatives to your specific application, process conditions, and stability requirements so you're not guessing your way through reformulation. #NaturalColorants #FoodFormulation #Spirulina #Confectionery #CleanLabel #DyeConverter

Will the color still be there in twelve months?
Will the color still be there in twelve months? Store lighting is the test most reformulations skip. The bottle on the left is the approved shade. The one on the right is the same formula after a year under retail lighting. Same product. Same recipe. The only variable was time under the lights. Natural pigments are photosensitive in ways synthetic dyes were not. Anthocyanins, betalains, and carotenoids absorb light and slowly break down. Fluorescent and LED case lighting runs the full life of the product on shelf. Bench-pass is not shelf-pass. This is why a color that looked perfect at sign-off can drift pale by the time a shopper reaches for it. The failure does not show up in the lab. It shows up in the store, months later, where nobody is watching. DyeConverter™ models light stability across the full shelf-life window, so the shade that gets approved is the shade that survives the lighting it will actually live under. Color is not a moment. It is a duration. #NaturalColorants #DyeConverter #CleanLabel

Before Red 40, there was hibiscus
Before Red 40, there was hibiscus. Before Yellow 5, there was marigold. Before Blue 1, there was butterfly pea flower. The plants that made the world colorful were doing it long before chemistry caught up. Petals. Roots. Bark. Algae. Insects. Spices. Every color a kid points at in a candy aisle started in a flower bed somewhere. Synthetic dyes didn't invent color. They just made it cheap, shelf-stable, and fast to source. Now the math has flipped. Synthetics are losing regulatory ground. Naturals are getting their second turn. And the question for every CPG team isn't "if" but "which one, in what dosage, in what matrix." That's where DyeConverter™ comes in. 15,000 natural ingredient swaps. 1 million data points. Every plant, every pigment, every pH window, every stability profile, indexed and ready. Stop waiting. Start shipping. #NaturalDyes #DyeConverter #FDA2027

DyeConverter™ doesn't save you money on ingredients
DyeConverter™ doesn't save you money on ingredients. Naturals still cost more per kilo than synthetics. That fight isn't winnable on raw cost. Where the savings actually live: BEFORE the bench. 78% faster reformulation time, because the platform models matrix compatibility before you commit to a candidate. 63% fewer bench tests, because you walk in with two or three high-confidence options instead of eight blind ones. 64% faster time to market, because supplier vetting, regulatory pre-screen, and CIELAB match scoring happen on screen, not on the phone. Regulatory risk: low. EU and FDA compliance checked before you order a sample, not after a failed scale-up. Estimated annual savings on a single SKU reformulation: $312K. Real money. Real ROI. Not a hoax. This is what onboarding in Q2 actually buys you. Not just a finished product on shelf, though that comes too. It buys you the math that makes 2027 survivable. Stop waiting. Start shipping. #NaturalDyes #DyeConverter #FDA2027

Q2 to Q4 sounds impossible
Q2 to Q4 sounds impossible. I hear you. If we're using the old reformulation playbook, it IS impossible. That playbook gave you time... time to research your options, time to get the samples in, time to test, time to fail and try again. That's how R&D used to budget for ingredient reform. The 2027 mandate didn't just shorten the timeline, it removed the space to fail. Every bad batch is real money. Every delayed sample request is real shelf space. Every sales call with a new supplier is a week you don't have. That's why our solution is BEFORE the bench. DyeConverter™ is built to shorten research time, mitigate supplier catalog confusion, and remove the guesswork. Tell us your matrix: your pH, your processing temps, your packaging, shelf life... even your cost ceiling. The platform models which natural dyes can meet those constraints. Each result has its own deep dive -- revealing confidence levels, conversion rates, dosages, performance prediction, cost analysis and regulatory data from agencies across the globe. You can even request a sample directly inside the platform without picking up the phone. Weeks are shaved off pre-bench research. Failures are mitigated at the bench. Getting to market is in sight and suddenly, "ROI" doesn't sound like a hoax. That's how onboarding with DyeConverter in Q2 can mean shipping with clean color by Q4. Stop waiting. Start shipping. #NaturalDyes #DyeConverter #FDA2027

December 2027
December 2027. That's when Red 40, Yellow 5, Yellow 6, Blue 1, Blue 2, and Green 3 leave the shelf. California is even earlier. If you're running R&D for a brand that uses any of those, the reformulation calendar started yesterday. DyeConverter(TM) is the decision model for synthetic dye reform. Every variable that affects a natural color swap, modeled before you bench test. pH. Shelf life. Packaging. Dosage. Regulatory status across US, EU, and Canada. Cost impact at scale. You stop guessing. You start validating. We're live and onboarding. Scan the code. #FutureOfFood #DyeConverter #FDA2027

Most reformulation projects start with a sample request
Most reformulation projects start with a sample request. Three suppliers, three dyes, rush them to the bench and start testing. Three weeks and four revisions later, the answer to "which one wins?" is, "it depends." (It depends on what got validated -- or didn't -- before the samples were ordered.) This four-question gate shortens research time and tightens the front end of any color reformulation project: → STABILITY: Will this color survive the next 6 to 12 months under light, oxygen, and time? Bench-pass is not shelf-pass. → APPLICATION: Does the formula context (fat vs water, dispersion, other actives) match how this color was characterized? → PROCESS CONDITIONS: Will it survive the actual pH, thermal, and shear envelope -- not just the supplier's spec sheet? → PERFORMANCE TARGETS: Are acceptable shade, intensity, and shelf-life floor defined in writing BEFORE the first trial? When the metrics are right on the front end, bench work moves faster. But your real cheat sheet is DyeConverter™ -- the only platform built on a decision model that maps these four checkpoints to the right dye for your specifications. We've launched, we're live, and we're onboarding now. (Use the QR code above to visit the official site.) The right color deserves the right input. Find it with DyeConverter. #CleanColor #NaturalDyes #FDA2027

You take a sip from the development bottle on Tuesday morning and
You take a sip from the development bottle on Tuesday morning and watch the bottom layer settle in real time. By the time the cap is back on, the drink is two distinct phases. Emulsion failure. The team starts naming suspects. The emulsifier choice. The homogenization pressure. The hold temperature. The fill order. Sometimes it is the color. Natural pigments are not inert ride-alongs in your matrix. Anthocyanin extracts bring water and acid. Carotenoids carry oils that demand re-balancing the surfactant load. Phycocyanin is a protein that argues with every other protein in the system. Drop the wrong color into a borderline-stable emulsion and you don't get a colored drink. You get a separated one. I've watched a beverage team rebuild their entire emulsion stack to chase a separation issue that turned out to be the pigment introducing 6% water into the formula. The color was the variable nobody flagged. That's why I built DyeConverter™. So you can see the water-activity, ionic, and emulsion implications of a natural color BEFORE you swap it in. Not after you are watching the layers settle. In clean-label work, color is never just color. It is a system input. #CleanLabel #NaturalDyes #FutureOfFood

You walk back to the display case
You walk back to the display case. The cupcake that piped perfectly an hour ago is now sliding off the wrapper. What broke? The team starts naming suspects. The mixer? The fat ratio? The emulsifier? The shop temperature? Sometimes it is the color. Natural pigments don't just sit there looking pretty. They bring water with them. They shift pH. They interact with the emulsifier system in ways your synthetic dye never did. When the fat network falls out of balance, the frosting looks fine in the bowl and weeps an hour later on the cupcake. I've watched a culinary team spend three days chasing the wrong variable on exactly this failure. The pigment was the one nobody flagged. That's why I built DyeConverter™. So you can see the water-activity, pH, and emulsion implications of a natural color BEFORE you swap it in -- not after the cupcakes are weeping. Just remember: in clean-label work, the ingredient is rarely the problem. The mismatch is. #CleanLabel #NaturalDyes #FutureOfFood

Why am I getting five different shades on the same spec?
Why am I getting five different shades on the same spec? Short answer: the spec measures how STRONG the color is, not WHICH color it is. Imagine walking into a paint store and asking for "red." You'd get handed a fan deck with fifty different reds on it. Cherry. Brick. Cranberry. Tomato. Fire engine. Every single chip says "red." Yet none of them are the same red. That's what an E-value is doing with natural color powders. E10 is basically a strength rating. It says how much color punch is packed into a gram of powder. It locks the INTENSITY. It does not lock the HUE. Why can five different "E10" powders look this different? Because the plant matters. An "E10 anthocyanin" might come from black carrot, purple corn, elderberry, red cabbage, or hibiscus. They're all in the red-to-purple family, but each plant's pigment is chemically a little different. Same family. Different cousins. Then there's the harvest year. The extraction method. The pH of whatever your color eventually lands in. All of it shifts the final shade without touching the E10 number on the certificate. This is why synthetic dyes felt so easy. Red 40 is Red 40, one molecule, one color, always. Natural dyes come from whole plants, and plants don't come with a part number. What helps: pick your supplier by the plant source, not just the E-value. Match color in your actual product, not in a water sample. And leave a little breathing room in your spec for batch-to-batch drift. Because that drift is not a defect; it's just what plants do. But if you want to mitigate bench time, start with DyeConverter™ *before* you even order that sample. Our platform will match you to the right SKUs based on your unique processing, pH and packaging data. Shade, source and supplier. No bias. Just the specs. Keep converting. #E10 #NaturalDyes #DyeConverter

Why is turquoise fading to green in my frozen popsicle?
Why is turquoise fading to green in my frozen popsicle? Short answer: spirulina blue isn't a pigment. It's a protein. And freezing damages proteins. The blue in spirulina comes from phycocyanin -- a phycobiliprotein, meaning a chromophore called phycocyanobilin sitting on a water-soluble protein scaffold. That scaffold is what holds the chromophore in the right position to absorb red-orange light and reflect blue. When the scaffold deforms, the color shifts. When a product containing phycocyanin freezes, ice crystal formation physically disrupts that protein structure. The chromophore starts to rotate, release, or aggregate. The wavelength being absorbed shifts. The color you see drifts from turquoise toward green, then yellow-green, most visibly at the edges and surfaces of the product where freezing stress is highest. This is different from how most other colorants behave. Synthetic dyes like Blue 1, and most natural pigments like anthocyanins and carotenoids, are small, stable molecules. Their color comes from structural chemistry, not from folded biology. You can freeze them, thaw them, refreeze them. The color holds. Phycocyanin also degrades in two other common process conditions. Above roughly 60°C the protein denatures and precipitates out of solution. Below pH 6 it unfolds and aggregates, clouding the blue. So frozen sorbets, acidic sports drinks, pasteurized smoothies -- anything that touches one of those zones -- is working against phycocyanin by default. A few things help. Fast freezing (blast freezing, liquid nitrogen) produces smaller ice crystals and less mechanical damage than slow freezing. Buffering toward neutral pH slows the acid-driven unfolding. Encapsulation in maltodextrin or gum arabic can physically protect the protein during processing. Some formulators also pair phycocyanin with stabilizers like xanthan gum to slow the visible fade, though this changes mouthfeel and neither are very 'clean label.' I've been in these quandaries too. I've been in "teams" of researchers, developers, food scientists and new product managers. I've also been the ONE "culinary specialist" on staff trying to make sense of things that are far beyond my pay grade. That's why I (personally) built DyeConverter™. Because the 2027 deadline isn't going anywhere, and we could all use a little help at the starting line with this one. Just remember, with natural dyes it's often not the ingredient causing the problem, it's the process. #CleanLabel #NaturalDyes #FutureOfFood
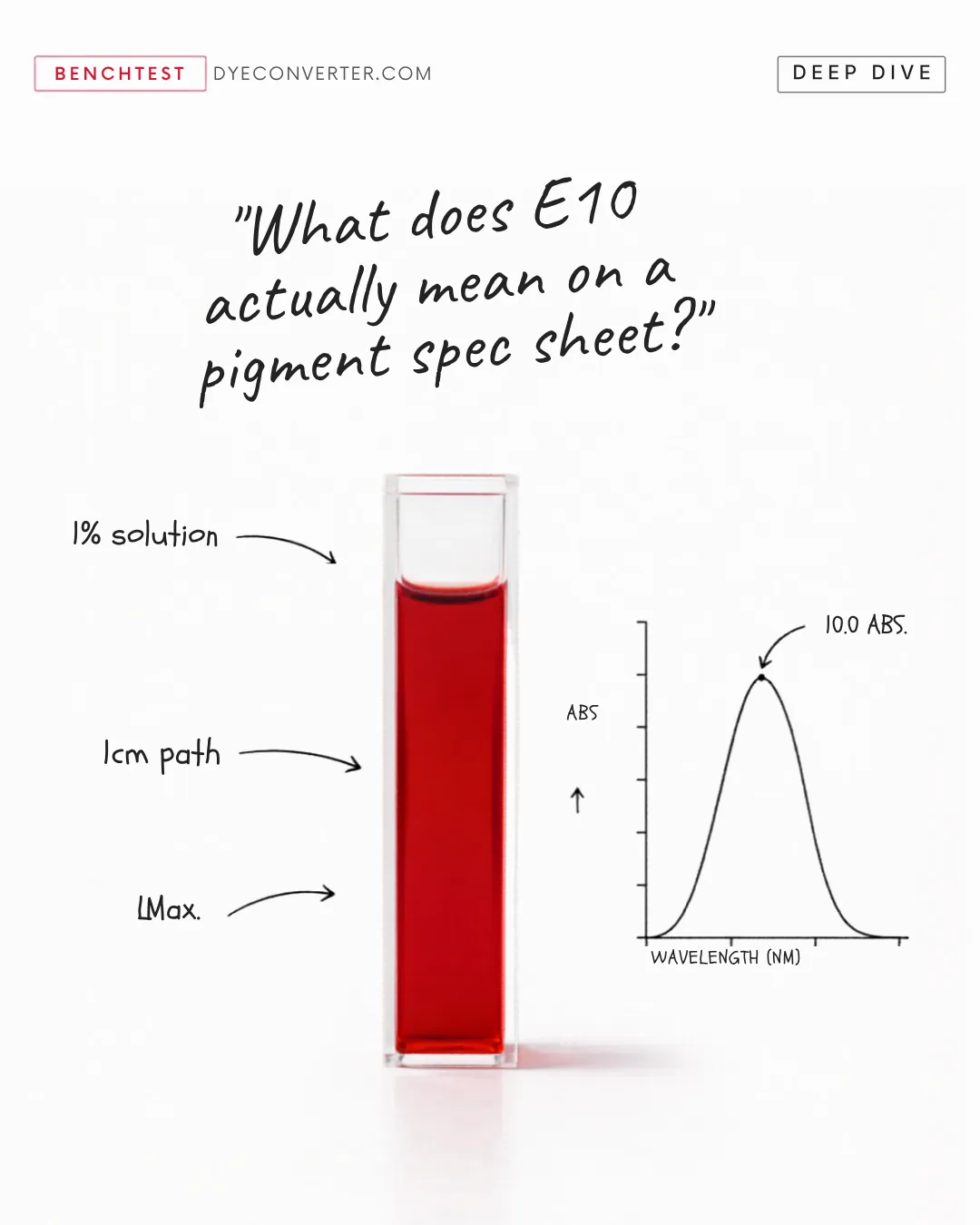
E10 might be the most useful, most misread number on a pigment spec
E10 might be the most useful, most misread number on a pigment spec sheet. Here's what it actually measures: the absorbance of a 1% pigment solution, read through a 1cm path. At the color's peak wavelength. Plain language? Color STRENGTH. How much pigment punch sits in a gram of your supplier's powder. Think of it like proof on alcohol -- tells you potency, not personality. The catch: two batches both certified E10 can land in your product looking noticeably different. The spec checks intensity. Hue rides along for free. That's why "same SKU, same E10" reformulations still drift between lots. The spec sheet was never measuring what your eye is. For natural colors, this isn't a defect. It's the material talking. The win is modeling the variation BEFORE it hits your line. DyeConverter™ predicts how a natural color will actually perform in your matrix -- pH, thermal, light, shelf life. The things a spec sheet doesn't tell you. Color strength is one number. Color truth is several. #FutureOfFood #DyeConverter #NaturalColors

Your dark beer looks great leaving the kettle
Your dark beer looks great leaving the kettle. By the time it's on the shelf, there's sediment at the bottom and haze you can't explain. This is one of the most common color defects in brewing. And it has nothing to do with filtration. Caramel colors are colloidal. They carry an electrical charge. Class 4 caramel has a strong negative charge. Beer proteins from malt carry a positive charge. Opposites attract. Literally. The negatively charged color particles bind to the positively charged proteins. They clump. They precipitate. That's your sediment. The technical fix is a Class III caramel, which carries a positive charge. But in a #CleanLabel world, "Ammonia Caramel" isn't the ingredient list your customers want to see. The real solution? Use the matrix to color the matrix. By swapping synthetic caramels for specialty malt extracts or caramelized sugar syrups, you achieve two things: Ionic Harmony: These plant-based concentrates are naturally compatible with beer proteins. No clumping. No sediment. Transparent Labeling: You trade an "E-number" or "Caramel Color" for "Malted Barley" or "Cooked Sugar." Stability doesn't have to come at the cost of your ingredient deck. DyeConverter™ is built to help you navigate these trade-offs -- matching ionic stability with clean-label demands before the shelf tells you something went wrong. #futureoffood #cleanlabel #foodfacts

BenchTest -- How do I know my paprika will stay fiery red in my corn
BenchTest #005 -- How do I know my paprika will stay fiery red in my corn chips for 12 months? (without waiting a year) Short answer: you cheat. Scientifically. It's called accelerated stability testing -- and it's one of the most underused tools in natural color reformulation. Instead of sitting around for 12 months hoping your paprika doesn't bail on you, you store your samples at elevated temperatures (around 32C / 90F) for 2 to 3 months. The heat fast-forwards degradation, giving you a reliable preview of how that pigment will hold up on a real shelf, in a real store, under real fluorescent lighting. Think of it like a time machine for your color. Minus the DeLorean. Here's what most R&D teams get wrong: they test in ideal conditions. Controlled humidity. Perfect darkness. Lab-grade packaging. That's not a Walmart shelf. Real-world testing means replicating the chaos -- temperature swings during shipping, light exposure in the store, oxygen ingress through packaging. Your paprika might look gorgeous in the lab and ghost you in the warehouse. The fix? Run your accelerated tests across multiple conditions. Light vs. dark. Ambient vs. stressed heat. Open vs. sealed. Three months of data at 32C will tell you more than a year of wishful thinking at room temp. And if the paprika doesn't make it? That's not a failure. That's intelligence. Now you know to explore a more heat-stable carotenoid or consider encapsulation before your product hits the line -- not after 50,000 units are already on trucks. The mandate isn't waiting. Neither should your stability data. #FutureOfFood #DyeConverter #BenchTest

Why is spirulina bleeding out of your white chocolate bunny?
Why is spirulina bleeding out of your white chocolate bunny? Phycocyanin -- the blue pigment in spirulina -- is water-soluble. White chocolate is a fat-based system. The pigment cannot bind into the fat matrix. It migrates to the surface and pools wherever moisture collects. This is the same reason liquid natural colors cause white chocolate to "seize." The water in the color dissolves sugar crystals, binds cocoa particles into a thick paste, and ruins the texture completely. The fix depends on format. Oil-soluble color dispersions or micronized powders integrate into the fat matrix without introducing water. But spirulina specifically is protein-based and heat-sensitive -- it denatures above 77C and degrades in acidic environments. For confections, the formulation window is narrow. DyeConverter™ maps which natural blue alternatives survive your specific process conditions -- heat profile, pH range, fat content -- so you stop discovering failures at the production line. #DyeConverter #CleanColor #FutureOfFood

Why does beet color speckle strawberry milk instead of turning it
Why does beet color speckle strawberry milk instead of turning it pink? Short answer: betanin does not dissolve in fat. And milk is full of it. Beet color gets its red from betanin, a water-soluble pigment. When you drop it into milk, the pigment dissolves into the water phase but cannot penetrate the fat globules. Those tiny fat droplets scatter light on their own, and the betanin clusters around them unevenly. The result is visible speckling instead of a smooth, uniform pink. Homogenization helps. Smaller fat globules mean more even pigment distribution. But even in highly homogenized milk, betanin can aggregate with casein proteins at certain pH ranges, creating micro-clumps that show up as darker pink spots. The real fix is particle size and dispersion method. Pre-dissolving the beet concentrate in warm water before blending into the dairy base gives the pigment time to fully hydrate and disperse before it hits the fat matrix. Some formulators also add a small amount of emulsifier to bridge the water-soluble pigment into a more uniform suspension. Temperature matters too. Betanin degrades above 50C and is most stable between pH 4 and 5. Milk sits around pH 6.7. That higher pH accelerates breakdown over shelf life, which is why beet-colored dairy products can shift from pink to brown faster than expected. This is one of dozens of dairy-specific formulation challenges mapped inside DyeConverter™. The platform matches natural color alternatives to your specific matrix, process conditions, and stability window so you stop troubleshooting by trial and error. #DyeConverter #CleanColor #FutureofFood